AI Radiology Software for Faster, Smarter Medical Imaging 4× Faster MRI Scans
An end-to-end radiology AI platform that accelerates MRI scans, automates reporting, and optimizes imaging workflows for hospitals and diagnostic centers.
Watch 1 min Solution Overview




Sites
One AI Platform.
Complete Radiology Workflow.
Aikenist combines PACS software, AI diagnostic tools, and workflow automation into a single platform designed to reduce reporting time, improve diagnostic accuracy, and increase imaging throughput without additional infrastructure.
AI-Powered Clinical Intelligence Across Specialties
Deploy advanced AI models for stroke detection, lung nodule identification, oncology imaging, and more.
Hospital-Grade Radiology Software Built for Scale
Designed for hospital IT systems and imaging centers with seamless HIS/RIS integration.
Transform Your Radiology Workflow with AI
See how Aikenist helps hospitals improve efficiency, reduce costs, and deliver faster diagnoses.

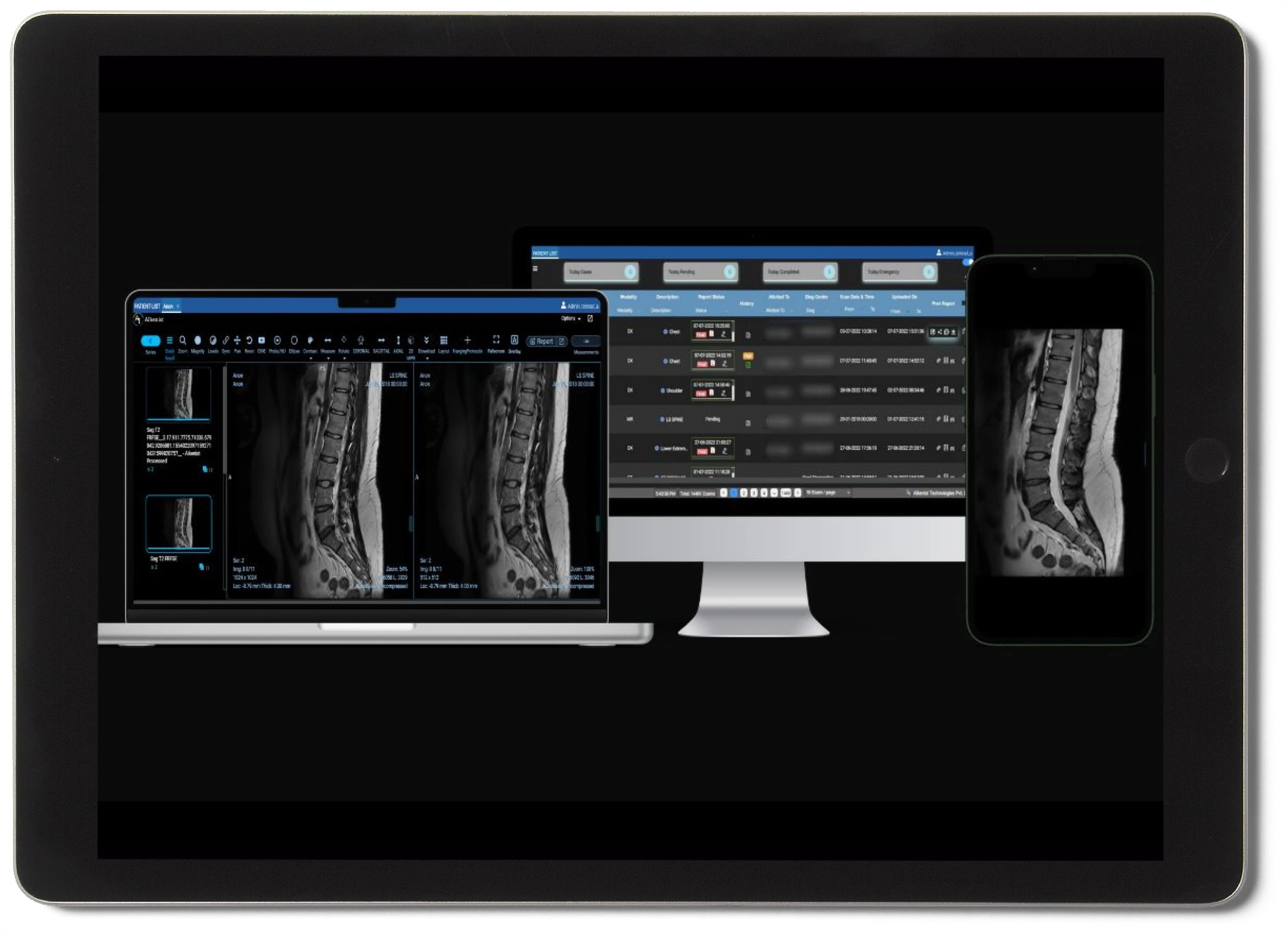
Eliminate manual bottlenecks across your entire radiology department with smart storing, intelligent reporting, and zero-footprint viewing — all in one agentic platform.
-
Agentic PACS & RIS
Next-generation AI-driven PACS that autonomously routes, tags, and prioritizes studies — reducing human effort at every step.
-
Zero-Footprint Smart Viewer
Browser-based viewer with 3D rendering, AI overlay annotations, and intelligent window/level presets for faster diagnosis.
-
Instant Collaboration
Real-time image sharing between radiologists, technicians, and referring physicians — across devices and locations.
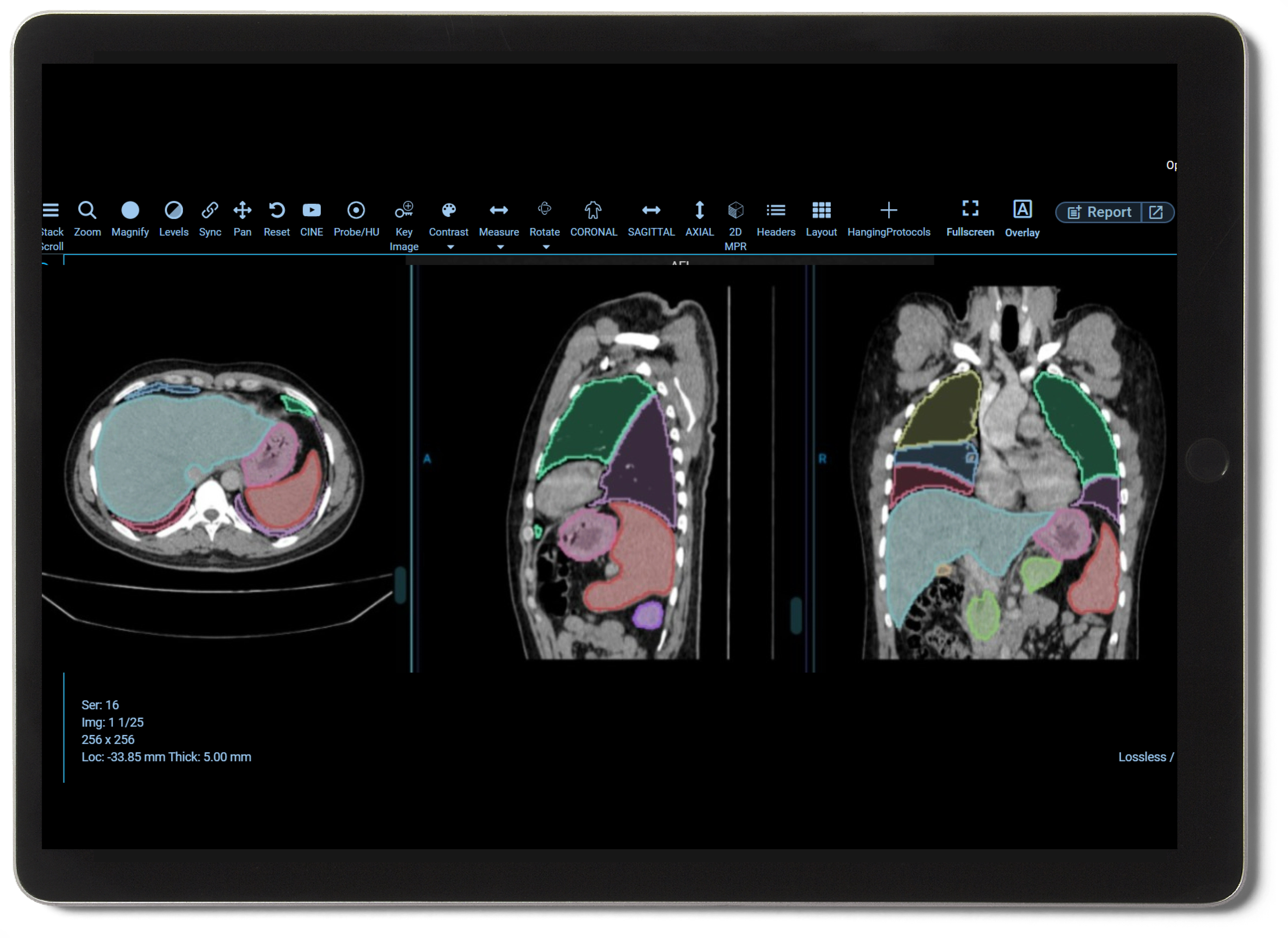

AI-powered diagnostic algorithms for Brain, Spine, Lung, and Abdomen — built for speed, precision, and critical-care workflows.
-
Multi-Organ AI Analysis
Specialized deep-learning algorithms for Brain, Spine, Chest, and Abdomen with sub-second inference and structured findings output.
-
Speech-to-Text Reporting
Convert spoken findings into structured, formatted radiology reports instantly — powered by medical-grade speech recognition. Cut reporting time by up to 60% without any typing.
-
Critical Alert Prioritization
Automatic triage and escalation for Stroke, TBI, Oncology, and Cardiology — ensuring time-sensitive cases reach the right hands first.

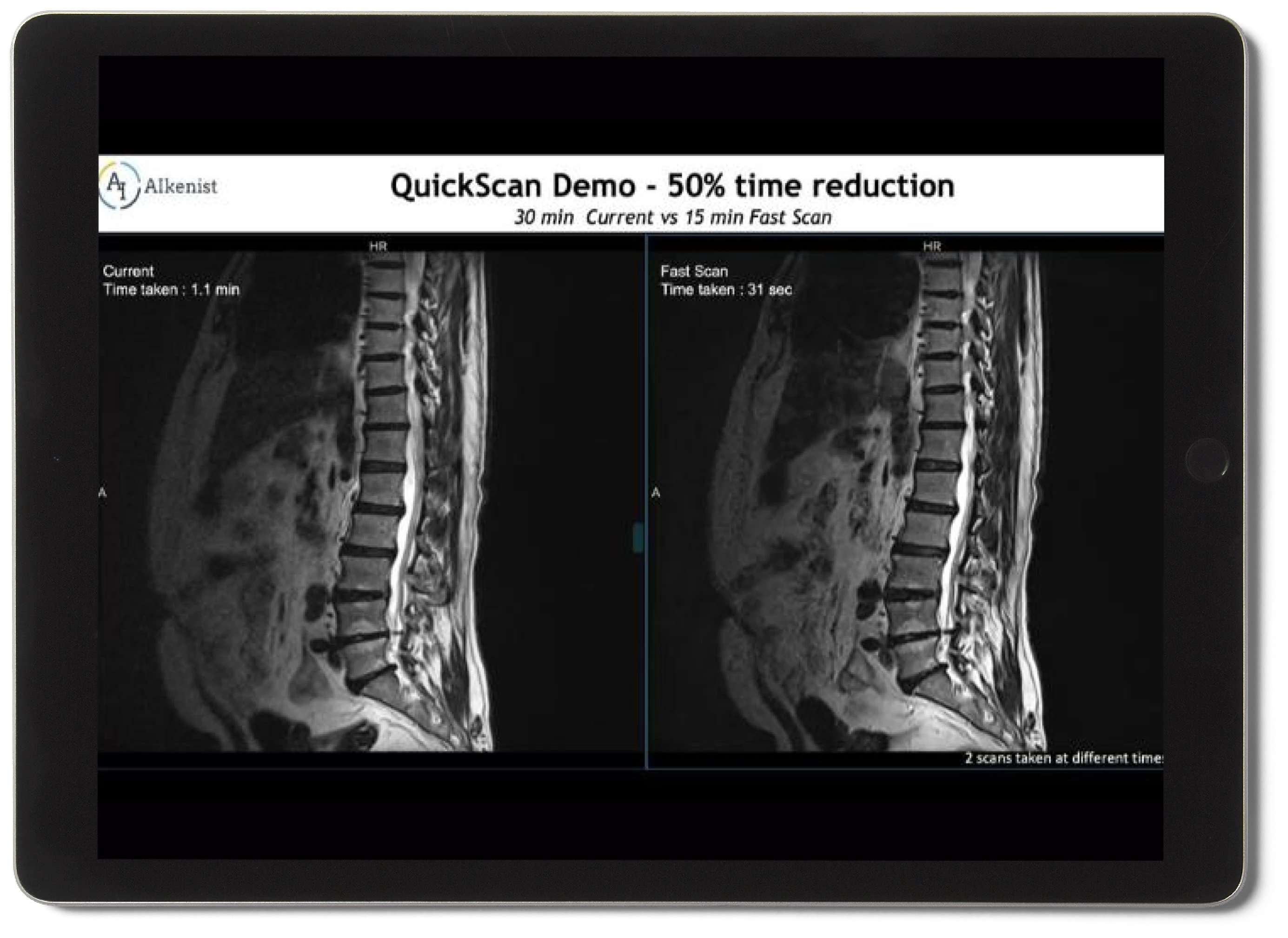
A software-only solution that accelerates MRI scans up to 4× — increasing throughput and reducing patient discomfort without any equipment changes.
-
4× Faster Scan Acquisition
Compress a 20-minute MRI down to 5 minutes — increasing throughput without adding hardware or staff.
-
Multi-Region Coverage
Supports Brain, Abdomen, Knee, Spine, and MSK scans across 1.5T and 3.0T field strengths.
-
AI Denoising & Enhancement
Deep learning reconstruction improves SNR and image sharpness — enabling better downstream AI detection accuracy.
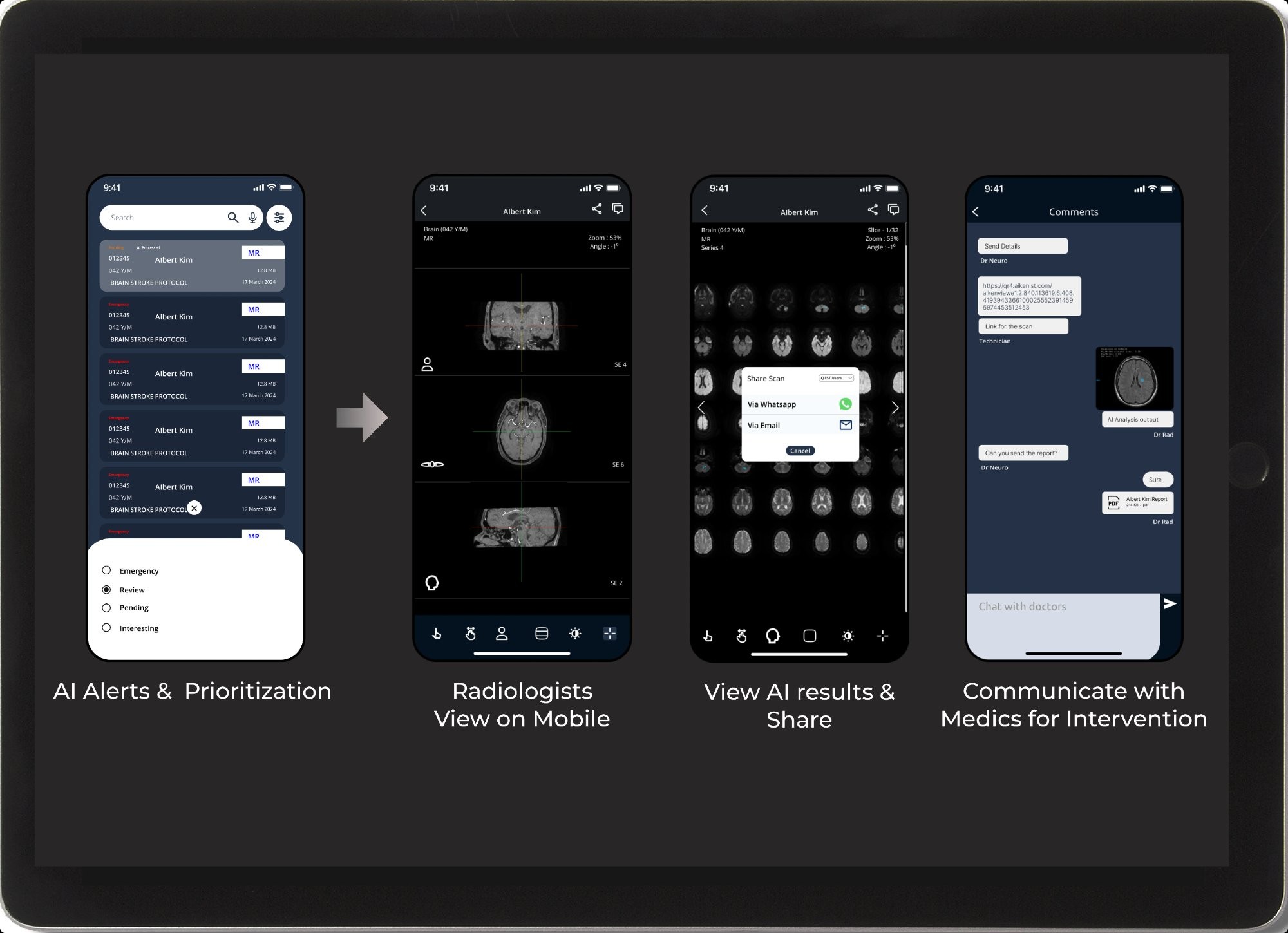

Manage every touchpoint of the patient's radiology journey — scheduling, check-in, scan, report, and delivery — all seamlessly integrated in a single AI-powered workflow.
-
End-to-End Patient Workflow
Online and offline booking, automated billing, and full traceability from appointment scheduling to final report delivery.
-
AI Queue Management
Intelligent slot optimization and patient prioritization for maximum scanner utilization and reduced wait times.
-
Emergency Fast-Track
Dedicated emergency pathways for Stroke, TBI, and COPD — auto-escalating critical cases for immediate care coordination.
Trusted by Radiologists & Hospitals
Hear directly from the clinicians, technologists, and administrators who use Aikenist every day.
Join 800+ Installations Across 10+ Countries
See how Aikenist can cut your
reporting time by 60% in 30 days.
Book Your Free Demo
Certified & Compliant Radiology Software
Every Aikenist product meets the highest global standards for medical device safety, data security, and clinical reliability.

FDA 510(k)
U.S. Food & Drug Administration
510(k) clearance confirms substantial equivalence to legally marketed predicate devices, enabling Aikenist AI tools to operate as Class II medical devices across the United States.

CDSCO
Central Drugs Standard Control Organisation
India's apex medical device regulator. Our MD-5 licence [MFG/MD/2024/000597] certifies Aikenist as a compliant AI diagnostic software manufacturer under Schedule M III quality standards.

ISO 13485
Medical Devices Quality Management System
The gold standard for medical device QMS. Validates our end-to-end quality processes — from R&D and manufacturing through post-market surveillance and risk management.

ANVISA
Agência Nacional de Vigilância Sanitária
ANVISA approval authorises Aikenist AI radiology products for Brazil's healthcare market — Latin America's largest — under RDC 751/2022 regulation for software as a medical device.

Korea MFDS
Ministry of Food and Drug Safety
Korea MFDS approval enables Aikenist AI products to be deployed across South Korean hospitals and imaging centres under the Medical Devices Act for AI-based diagnostic software.
Vulnerability Assessment & Penetration Testing
Independent security audit — annually renewed
Comprehensive third-party security audit covering network infrastructure, application layer, REST APIs, and database endpoints. Protects patient data against intrusion, SQL injection, XSS, and unauthorized access.
Indian Computer Emergency Response Team
Ministry of Electronics & IT, Government of India
Full compliance with CERT-In directives under India's IT Act 2000. Mandates timely cybersecurity incident reporting within 6 hours, 180-day log retention, synchronised system clocks, and mandatory audit trails.
Health Insurance Portability & Accountability Act
U.S. Department of Health & Human Services
Full compliance with HIPAA Privacy and Security Rules. All patient data is AES-256 encrypted at rest and TLS 1.3 in transit. Role-based access controls, audit logs, and signed Business Associate Agreements are enforced.
Digital Imaging & Communications in Medicine
NEMA Standard — Global Medical Imaging Interoperability
Native DICOM conformance ensures plug-and-play interoperability with any scanner, PACS, or RIS. Supports DICOM SR, WADO-RS, and HL7 FHIR for comprehensive health data exchange.
Information Security Management System
ISO/IEC 27001 — International Standard
ISO 27001 certification validates Aikenist's enterprise-grade information security management system. Covers risk assessment, access control, incident response, and continuous security improvement across all systems handling patient and clinical data.
Agência Nacional de Vigilância Sanitária
Brazil National Health Surveillance Agency
ANVISA approval authorises Aikenist AI radiology products for the Brazilian healthcare market — Latin America's largest. The clearance confirms compliance with Brazil's RDC 751/2022 regulation for software as a medical device (SaMD).
Ministry of Food and Drug Safety
Korea MFDS — Republic of South Korea
Korea MFDS approval enables Aikenist AI products to be marketed and deployed across South Korean hospitals and imaging centres. The clearance is issued under the Medical Devices Act and confirms safety and efficacy for AI-based diagnostic software.
European Conformity — Medical Device Regulation
EU MDR 2017/745 — Class IIa Medical Device
CE marking under EU MDR 2017/745 enables Aikenist products to be marketed across all 27 EU member states as a certified Class IIa AI-based medical device. Technical file under review by notified body.
All Aikenist products meet global medical device standards — ready to deploy in your institution today.
Book a Demo — See It Live View Full Compliance DetailsHow QuickSuite Works
A seamless AI-driven pipeline — from patient booking to final report — built on Cloud & DC infrastructure.
Booking & Registration
- Online/Offline Booking & Billing via AI
- RIS with HIS Integration
- Patient Portal
AI-Accelerated Scanning
- MRI: 2× Fast Scan, safer & more accurate
- CT: Low Dose CT acquisition
- 70% better Signal-to-Noise Ratio
Agentic PACS & Distribution
- AI PACS & RIS with smart routing
- Smart Workflow, Viewer & Teleradiology
- Instant sharing to Doctor & Patient
AI Diagnosis & Reporting
- AI reporting with Speech-to-Text synthesis
- Brain, Spine, Lung & Abdomen analysis
- StrokeSuite & OncoSuite pathways
Delivered
Experience this end-to-end workflow live in your own radiology environment.
Schedule a Workflow DemoAI + KEN = AI Knowledge Specialist
Backed by Research.
Proven in Practice.
Our work is grounded in peer-reviewed research, clinical validation, and real-world deployments across leading radiology centres.
Interoperable
Works seamlessly with multiple scanners and PACS systems out of the box.
Data Safe
Patient Health Information (PHI) anonymized and fully secured at every layer.
Easy to Deploy
Managed deployment at local data center or cloud — up and running fast.
Clinically Validated
Algorithms trained and validated on diverse, multi-centre clinical datasets.
Aikenist at ECR 2026
Excited to connect at the European Congress of Radiology (ECR) 2026. Aikenist is redefining radiology with one intelligent, agentic workflow.
Agentic Radiology WorkFlow OS
Patient. Radiologist. Clinician. Hospital. One Aikenist Technologies. One Agentic Radiology WorkFlow OS. AI, powered by the knowledge of everyone it serves.
The Radiology AI Market Thesis
Pure algorithm vendors lose. Not because their technology is inferior, but because detection without orchestration is incomplete. The era of 'AI as feature' is over. The era of 'AI as operating system' is here.
Ready to bring clinically-validated AI to your radiology department?
Book a 20-Min DemoWatch QuickSuite Live
See how our AI-powered radiology platform performs in real clinical environments.
Trusted by Leaders
From global research institutions to leading diagnostic centres — Aikenist powers radiology across the ecosystem.
Associations & Partners
Global research bodies, accelerators, and strategic alliances
 C-CAMP
C-CAMP MARL5G
MARL5G CCMB
CCMB A*STAR
A*STAR MGH
MGH HLL Lifecare
HLL Lifecare Jio GenNext
Jio GenNext Hayleys Fentons
Hayleys Fentons Dell
Dell Toprad
Toprad HCKonnect
HCKonnectIndia
Hospitals, diagnostic chains, and imaging centres across India
 Atlantis
Atlantis Jaydeva
Jaydeva Kidwai
Kidwai JSS
JSS Malla Reddy
Malla Reddy Victoria
Victoria Command Hospital
Command Hospital Star Hospitals
Star Hospitals Prakriya
Prakriya Pi Health
Pi Health Isha
Isha Manipal Health
Manipal Health SGVP
SGVP Medicover
Medicover Medall
Medall Mahajan Imaging
Mahajan Imaging Mediscan
Mediscan Konnect
Konnect Aarthi Scans
Aarthi Scans Nirmal
Nirmal Accurate
Accurate Addannex
Addannex GenX
GenX Prima
Prima Primescan
Primescan Raddoc Live
Raddoc Live RRS
RRS Saral
Saral X-Ray House
X-Ray House GTR
GTRInternational
Global deployments across the UK, US, and Africa
 Chartwell UK
Chartwell UK Radblox US
Radblox US Clinix Nigeria
Clinix Nigeria Toprad US
Toprad US Cerebriu Denmark
Cerebriu DenmarkCommunication
Messaging and notification integrations for patient and radiologist workflows
 Twilio
Twilio WhatsApp
WhatsApp OsiriX
OsiriX Sectra IEP
Sectra IEP Radiant Dicom Viewer
Radiant Dicom ViewerHIS / LIMS
Hospital information and lab management system connectors
 EPIC
EPIC eClinicalWorks
eClinicalWorks Sufalam
Sufalam IT Dose
IT Dose Akhil Systems
Akhil SystemsAI
Artificial intelligence and decision support tools embedded in the reading workflow
 Qure.ai
Qure.ai Cerebriu
Cerebriu Azmed
Azmed Carpl
Carpl HCKonnect
HCKonnect3D Workstation
Advanced volumetric rendering and 3D visualisation platforms
 TeraRecon
TeraRecon Inobitec
InobitecHardware
High-performance computing hardware optimised for medical imaging workloads
 NVIDIA
NVIDIA Dell
DellJoin 800+ installations from world-class institutions who trust Aikenist for their radiology workflows.
Schedule Your Demo TodayThe Team Behind Aikenist
A multidisciplinary team of engineers, clinicians, and researchers building the future of radiology AI.
To improve clinical response time, patient convenience, and resource utilisation. Aikenist is a healthcare technology company focused on designing products that enhance the end-to-end patient experience and optimise the utilisation of medical equipment.




Book a Demo
Schedule a personalized demo to explore how Aikenist improves efficiency and diagnostics.
See QuickSuite running in a real radiology environment — not a slide deck.
Tell us your modalities, scanner models, and pain points — we'll demo exactly what matters to you.
A focused session with a clinical specialist. We respond within 4 business hours.
Fill in the details below and a clinical specialist will reach out within 4 business hours to confirm your slot.
Log in to upload scans, view reports & manage your workflow.